- 5-year survival rate of 82.6% and 73% reduction in overall risk of death (hazard ratio of 0.27) are the strongest Multikine efficacy results reported to date, reflecting results in patients who met the CEL-SCI confirmatory Registration Study target population criteria and were deemed at low risk for recurrence, therefore not recommended per NCCN guidelines to receive chemotherapy post-surgery
- Data provide further evidence that the selection criteria for CEL-SCI’s upcoming confirmatory Registration Study may yield improved clinical outcomes
- Future advances in diagnostic technologies that improve patient selection, such as PET scans (which will be used in the confirmatory study), may further optimize patient selection and Multikine’s already strong efficacy results from the current 5-year Overall Survival at 73% up into the 82% range
- Clear unmet need: the 5-year survival rate remains below 50% in locally advanced resectable head and neck cancer patients who receive standard of care only
CEL-SCI Corporation (NYSE American: CVM) today reported new data from its concluded Phase 3 study of Multikine® (Leukocyte Interleukin, Injection)* that were presented at the European Society for Medical Oncology (ESMO) 2024 Congress on Saturday, September 14, 2024 in a poster titled “Prognostic significance of diagnostic staging in treatment naïve, resectable locally advanced primary oral cavity squamous cell carcinoma for neoadjuvant Leukocyte Interleukin Injection immunotherapy”. This data is highly relevant to CEL-SCI’s 212 patient confirmatory Registration Study which has received the U.S. Food and Drug Administration’s (FDA) go-ahead and is currently under preparation.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20240916023296/en/
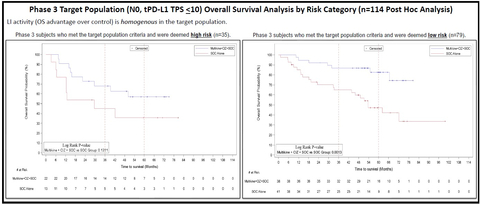
CEL-SCI’s Multikine® Increases Head and Neck Cancer 5-Year Survival Rate to 82.6%. The graph on the right side (n=79) demonstrates that the target low risk population had a 5-year survival rate of 82.6% when treated with Multikine (n=38) vs. 47.3% when treated with standard of care alone (n=41). (Graphic: Business Wire)
Summary of Phase 3 Study: Multikine-treated patients who were recommended treatment of surgery and radiotherapy had a nearly 4-year survival benefit over control group
As previously reported, CEL-SCI’s completed Phase 3 study of 923 patients showed that newly diagnosed head and neck cancer patients who were deemed at low risk for recurrence after surgery (and therefore recommended to receive only radiotherapy after surgery) had a median overall survival (OS) benefit of 46.5-months, almost 4-years, over control patients. However, patients who were deemed to be high risk for recurrence after surgery (and therefore recommended to have chemotherapy added to the radiotherapy after surgery) showed no survival benefit.
Upcoming FDA Confirmatory Registration Study
Since the completed Phase 3 study showed clear survival benefit for some, but not all of the patients, the FDA requested that CEL-SCI conduct a confirmatory Registration Study focusing on the patients who showed the best survival benefit. Based on the data, CEL-SCI determined this target population to be patients with newly diagnosed locally advanced primary head and neck cancer with no lymph node involvement and with low PD-L1 tumor expression. Applying these selection criteria to the completed Phase 3 study of 923 patients resulted in the target population (n=114) having a 73% survival at 5 years vs a 45% survival at 5 years for the control patients, log rank p=0.0015. The hazard ratio was an exceptional 0.34, with a 95% confidence interval upper limit of 0.65; Wald p=0.0012 and achieving a 66% reduction in the overall risk of death.
Summary of New Data Presented at ESMO
The new data presented at ESMO includes a further analysis of the 114 patients in the completed Phase 3 study who met these target population selection criteria and form the basis for the confirmatory study. Specifically, the new analysis focused on those patients who were deemed low risk for recurrence (recommended to be given only radiotherapy – but no chemotherapy, per National Comprehensive Cancer Network “NCCN” guidelines) following surgery (n=79) as opposed to the selected patients who were deemed high risk for recurrence and who were recommended to have chemotherapy added to their treatment following surgery per the same guidelines (n=35).
While the overall survival benefit was clear and statistically significant (log rank p=0.0015) for the entire target population (n=114), the 79 patients who were recommended to receive only radiotherapy benefited to an even greater degree from pre-surgery treatment with Multikine than the group of 114 as a whole. This target low risk population (n=38) had a 5-year overall survival of 82.6% when treated with Multikine vs. 47.3% when treated with standard of care alone (n=41), without overlap in their respective 95% confidence intervals. More recent analysis for the target low risk population (n=79) showed a hazard ratio of 0.27 (95% CI [0.12, 0.64], Wald p=0.0027) achieving a 73% reduction in overall risk of death.
Management Commentary
“The additional data presented this weekend at ESMO 2024 provides further evidence that we have identified the target population that has the greatest survival benefit from Multikine, and that our study criteria can select for these patients upon diagnosis, before surgery,” stated CEL-SCI CEO Geert Kersten. “It makes sense that Multikine, an immunotherapy, provides even greater benefit to patients who are not scheduled to receive chemotherapy following surgery, given the known detriments of chemotherapy on the immune system. Seeing more clearly than ever that patients who were not recommended chemotherapy benefited the most begs the question: What if, through better diagnostic technology such as the PET scan, which we will be using in the confirmatory study, resulting in better patient selection, we could treat only those patients who are supposed to be treated with radiotherapy alone, and not chemotherapy? The data presented at ESMO is clear. This would lead to even better 5-year survival, 82% instead of 73%.”
CEL-SCI’s CSO Eyal Talor, Ph.D. commented, “The criteria we developed for selecting these locally advanced head and neck cancer patients clearly showed that when patients were treated with Multikine before surgery, they demonstrated an overall survival advantage over control irrespective of whether these patients were characterized as being at low- or high-risk for recurrence following surgery. With this new analysis we also saw that patients selected by these criteria who are deemed low risk for recurrence post-surgery have a further improved survival outcome with a hazard ratio of 0.27, which even is better than the already exceptional hazard ratio of 0.34 seen for the overall selected population.”
The data were presented at ESMO 2024 by the study’s co-author József Tímár MD, PhD, DSc, Professor Department of Pathology, Forensic and Insurance Medicine at Semmelweis University in Budapest, Hungary. Dr. Timar served as the Director of the Central Pathology Laboratory for CEL-SCI’s IT-MATTERS Phase 3 study. With 174 peer reviewed studies published, Dr. Timar is a founding editor, editor in chief, or a member of the editorial board of four oncology journals. He is the recipient of a dozen honors and awards for excellence in cancer research and teaching.
About CEL-SCI Corporation
CEL-SCI believes that boosting a patient’s immune system while it is still intact should provide the greatest possible impact on survival. Multikine is designed to help the immune system "target" the tumor at a time when the immune system is still relatively intact and thereby thought to be better able to mount an attack on the tumor.
Multikine (Leukocyte Interleukin, Injection), a true first-line cancer therapy, has been dosed in over 740 patients and received Orphan Drug designation from the FDA for neoadjuvant therapy in patients with squamous cell carcinoma (cancer) of the head and neck. Based on the very strong data from the completed randomized controlled Phase 3 study, the FDA agreed to CEL-SCI’s target patient selection criteria and gave the go-ahead to conduct a small, focused, confirmatory Registration Study which will enroll 212 patients. CEL-SCI will enroll newly diagnosed locally advanced primary head and neck cancer patients with no lymph node involvement (determined via PET scan) and with low PD-L1 tumor expression (determined via biopsy), representing over 100,000 patients annually.
The Company has operations in Vienna, Virginia, and near/in Baltimore, Maryland.
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. When used in this press release, the words "intends," "believes," "anticipated," "plans" and "expects," and similar expressions, are intended to identify forward-looking statements. Such statements are subject to risks and uncertainties that could cause actual results to differ materially from those projected. Factors that could cause or contribute to such differences include an inability to duplicate the clinical results demonstrated in clinical studies, timely development of any potential products that can be shown to be safe and effective, receiving necessary regulatory approvals, difficulties in manufacturing any of the Company's potential products, inability to raise the necessary capital and the risk factors set forth from time to time in CEL-SCI's filings with the Securities and Exchange Commission, including but not limited to its report on Form 10-K for the year ended September 30, 2023. The Company undertakes no obligation to publicly release the result of any revision to these forward-looking statements which may be made to reflect the events or circumstances after the date hereof or to reflect the occurrence of unanticipated events.
* Multikine (Leukocyte Interleukin, Injection) is the trademark that CEL-SCI has registered for this investigational therapy. This proprietary name is subject to FDA review in connection with the Company's future anticipated regulatory submission for approval. Multikine has not been licensed or approved for sale, barter or exchange by the FDA or any other regulatory agency. Similarly, its safety or efficacy has not been established for any use.
View source version on businesswire.com: https://www.businesswire.com/news/home/20240916023296/en/
Contacts
Gavin de Windt
CEL-SCI Corporation
(703) 506-9460













